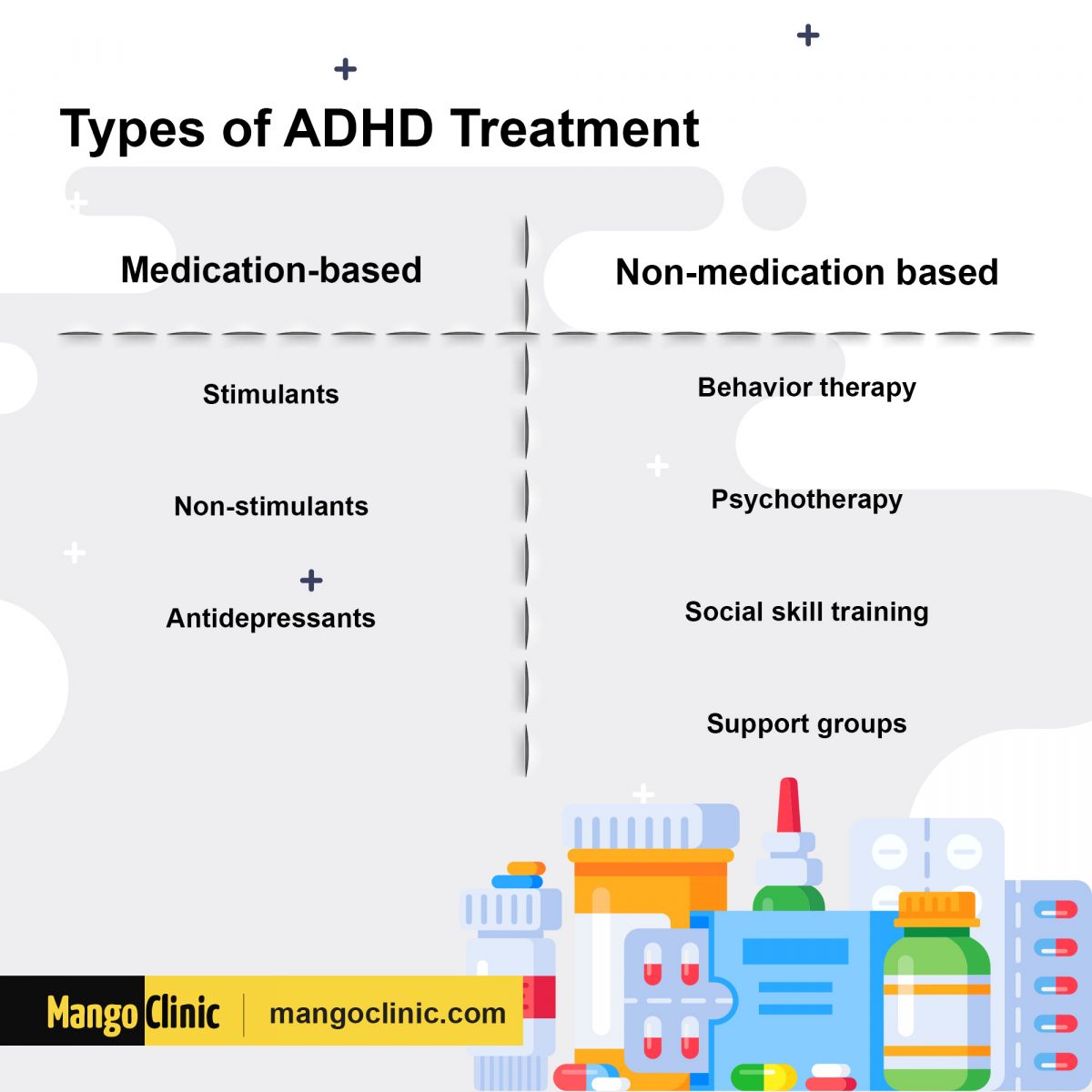
The eTNS effects are bigger than those found in other studies for non-drug therapies like neurofeedback, child and parent training, omega-3 fatty acids, and cognitive therapy. While eTNS hasn’t been compared head-to-head with other ADHD treatments, Loo notes that the improvements are on par with those of the widely-used, non-stimulant ADHD medications atomoxetine and guanfacine.īut stimulant ADHD drugs are generally regarded as more effective. How Does eTNS Compare to Other Treatments for ADHD? They may feel a slight tingling sensation on their skin. A wire sends low-level electrical pulses to an adhesive patch placed on the child’s forehead, just above their eyebrows. Kids wear the eTNS unit at night, clipped to their pajamas. ²It was the agency’s first OK of a non-drug device for the treatment of ADHD. In the spring of 2019, the US Food and Drug Administration approved the marketing of the prescription-only device for children ages 7 to 12 with ADHD who are not taking medications for the condition. The study was published in the April 2019 issue of the Journal of the American Academy of Child & Adolescent Psychiatry. In comparison, just 14% in a control group saw improvements. 
In 2019, a small University of California Los Angeles study of 62 girls and boys, ages 8 to 12, with moderate to severe ADHD found that 52% of those treated with the Monarch external Trigeminal Nerve Stimulation (eTNS) System for four weeks had significant reductions in inattentiveness, hyperactivity, and impulsiveness.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
